搜索
品名
结构式
CAS号码
质量标准
适应症
状态
阿帕他胺
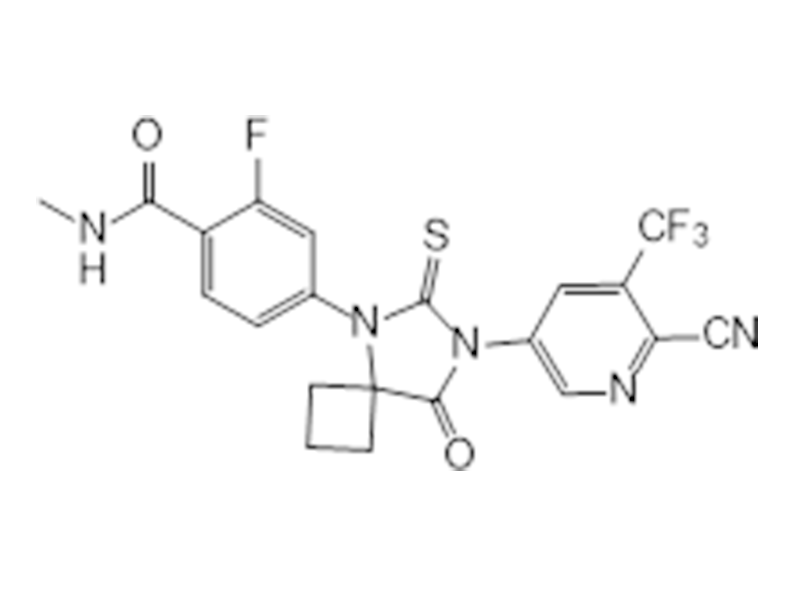

956104-40-8
企标
抗癌
商业化/验证完成
阿哌沙班
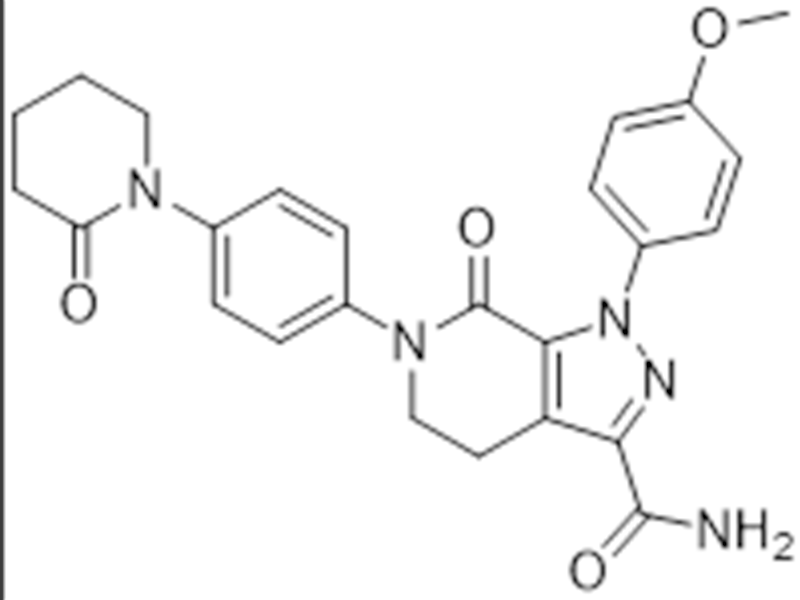

503612-47-3
企标
抗凝血
商业化/验证完成
马来酸阿伐曲泊帕
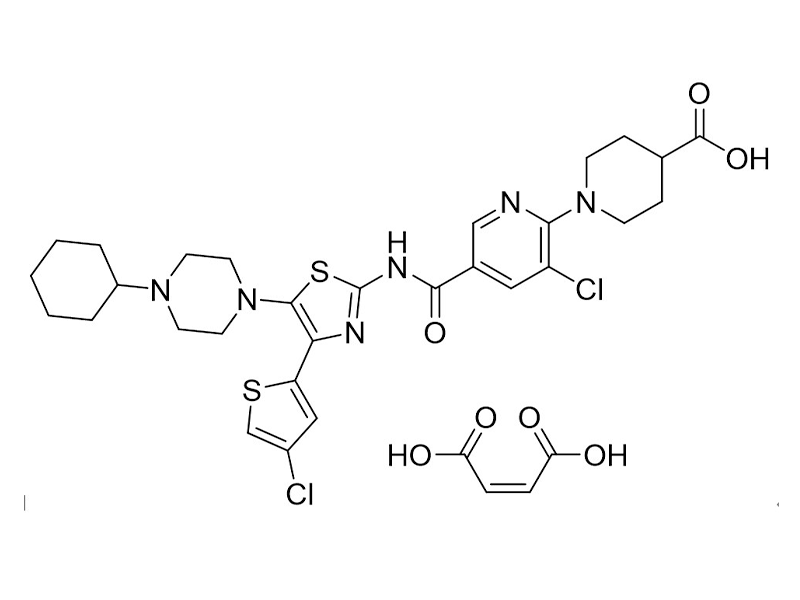

570406-98-3
企标
抗血栓
商业化/验证完成
阿齐沙坦
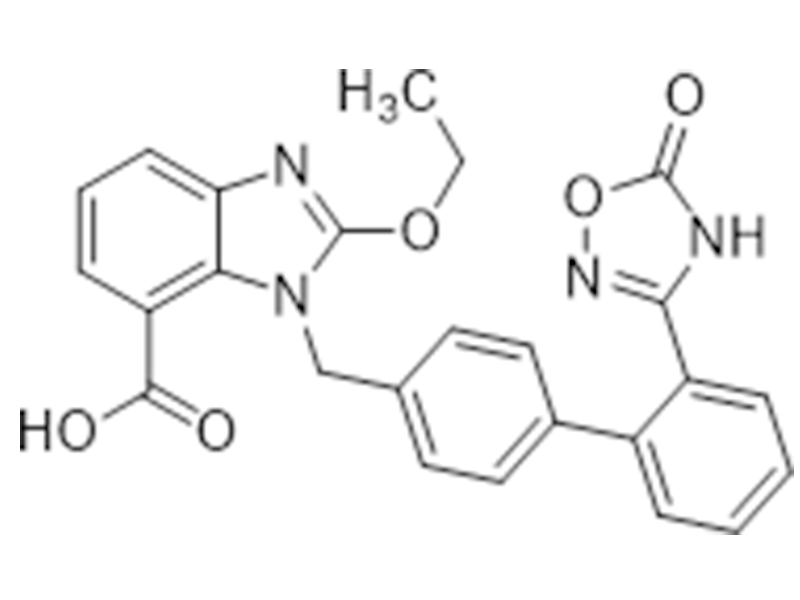

147403-03-0
企标
抗高血压
商业化/验证完成
美阿沙坦钾
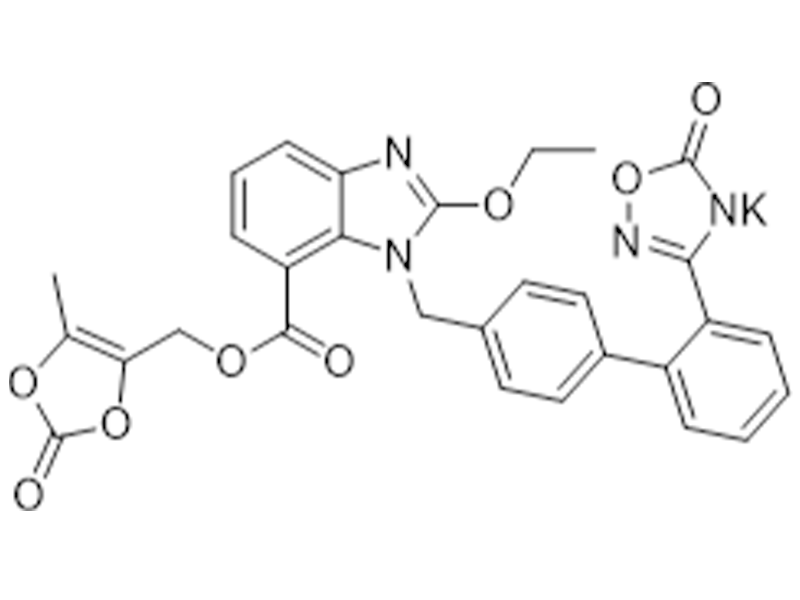

863031-24-7
企标
抗高血压
商业化/验证完成
布瑞哌唑
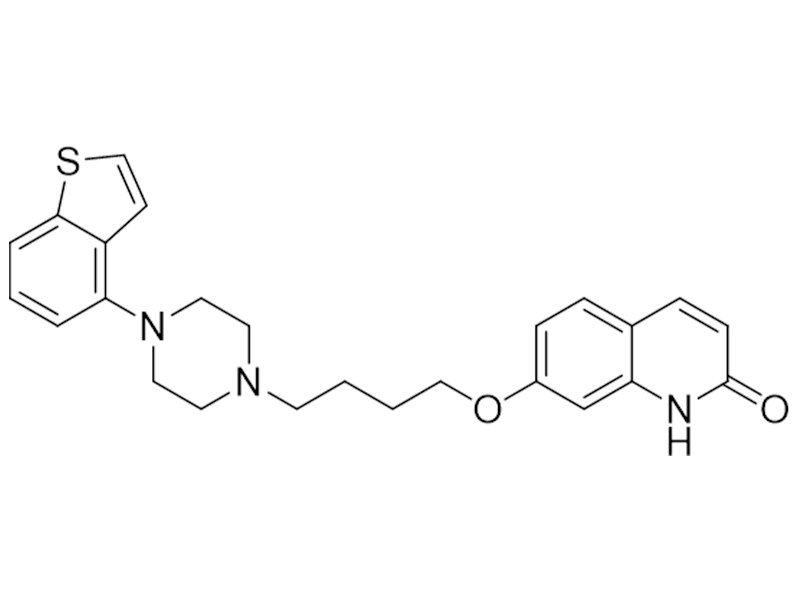

913611-97-9
企标
抗抑郁
商业化/验证完成
布立西坦
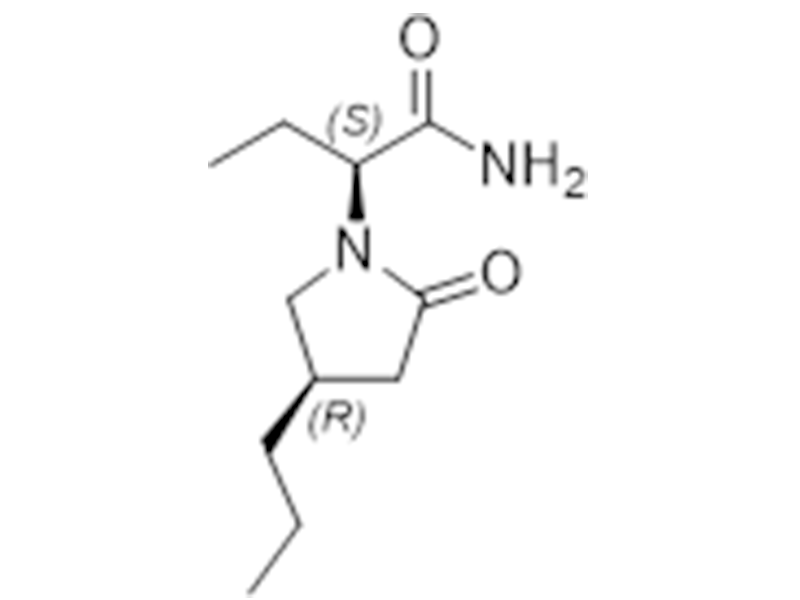

357336-20-0
企标
抗癫痫
商业化/验证完成
苹果酸卡博替尼
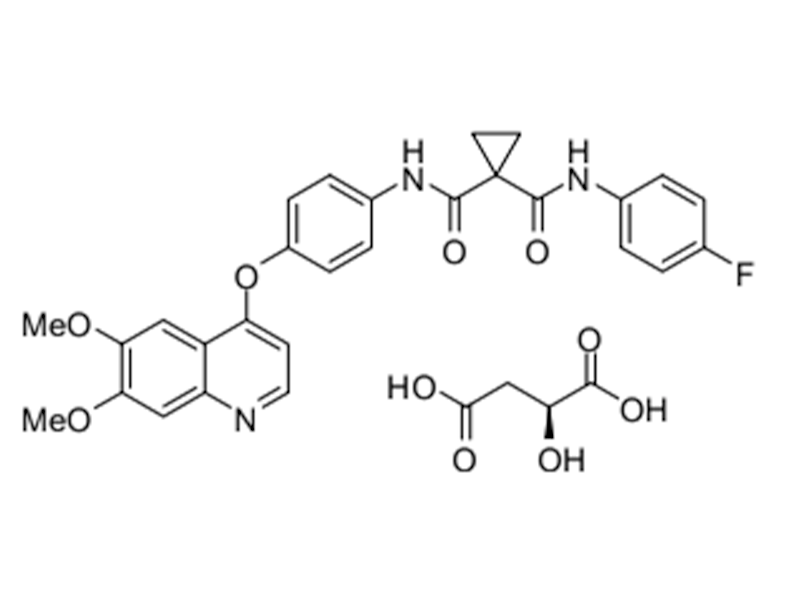

1140909-48-3
企标
抗癌
商业化/验证完成
卡格列净半水合物
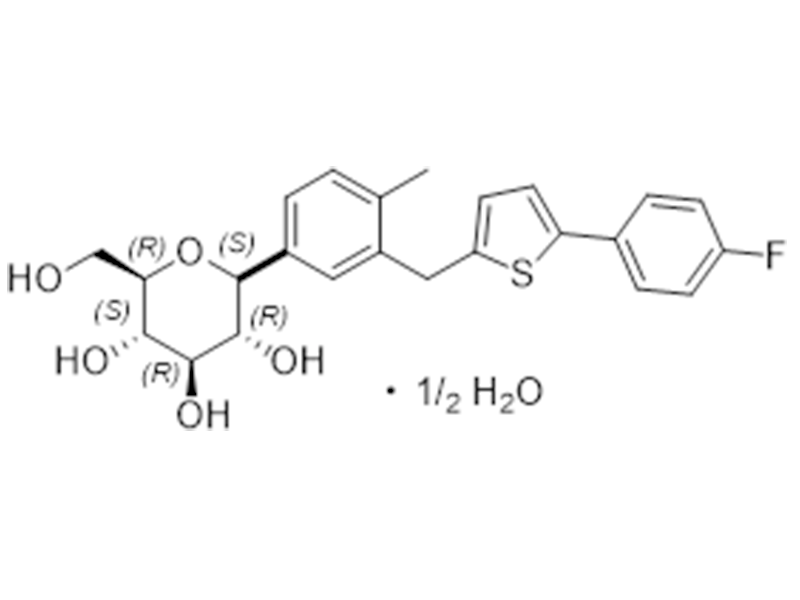

928672-86-0
企标
SGL2抑制剂/抗糖尿病
商业化/验证完成
坎地沙坦酯
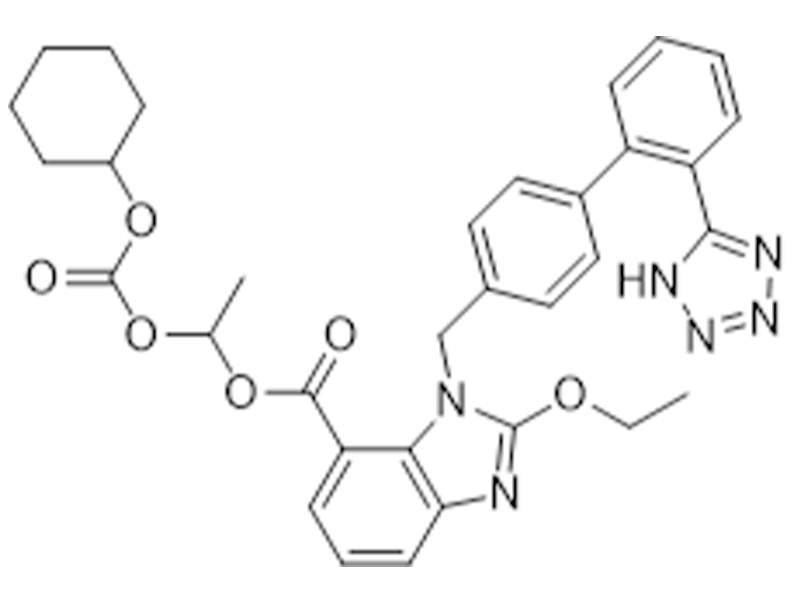

145040-37-5
USP/EP
抗高血压
商业化/验证完成
甲磺酸达比加群酯
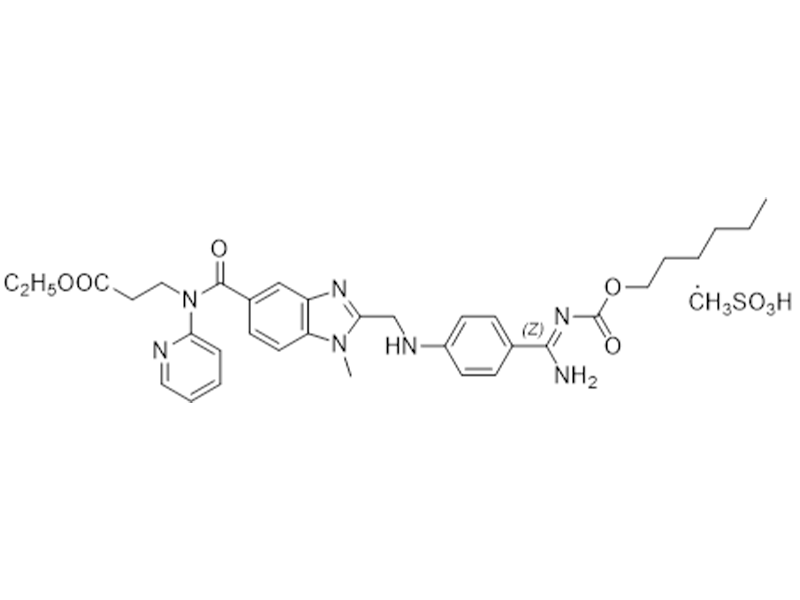

872728-81-9
企标
抗凝血
商业化/验证完成
达格列净
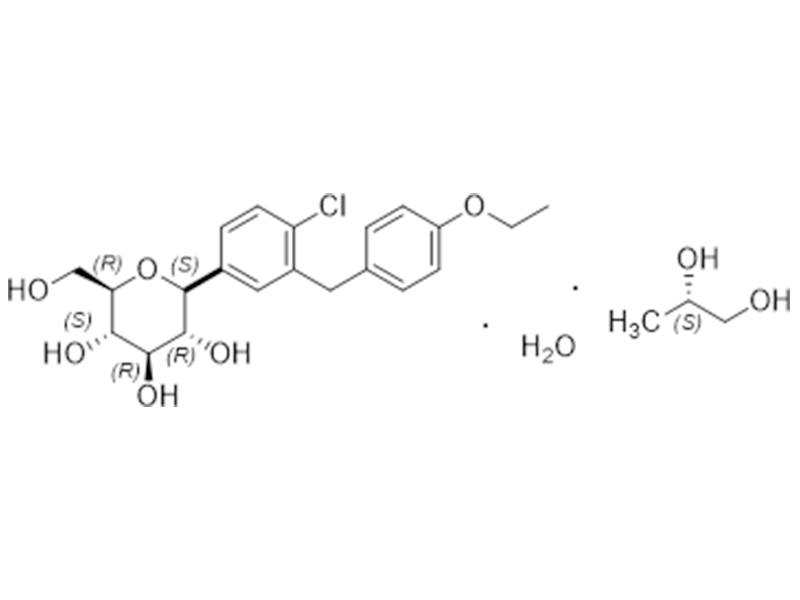

960404-48-2
企标
SGL2抑制剂/抗糖尿病
商业化/验证完成
艾多沙班对甲苯磺酸一水合物
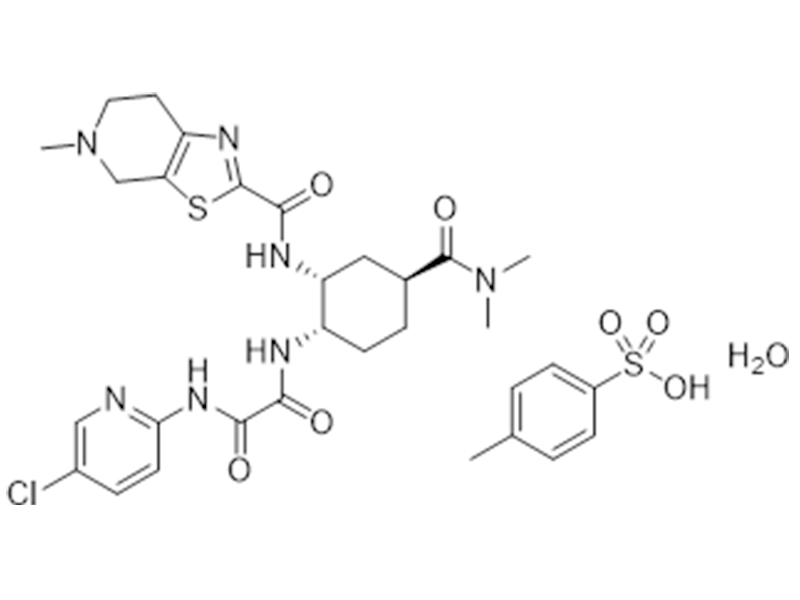

1229194-11-9
企标
抗凝血
商业化/验证完成
噁拉戈利钠
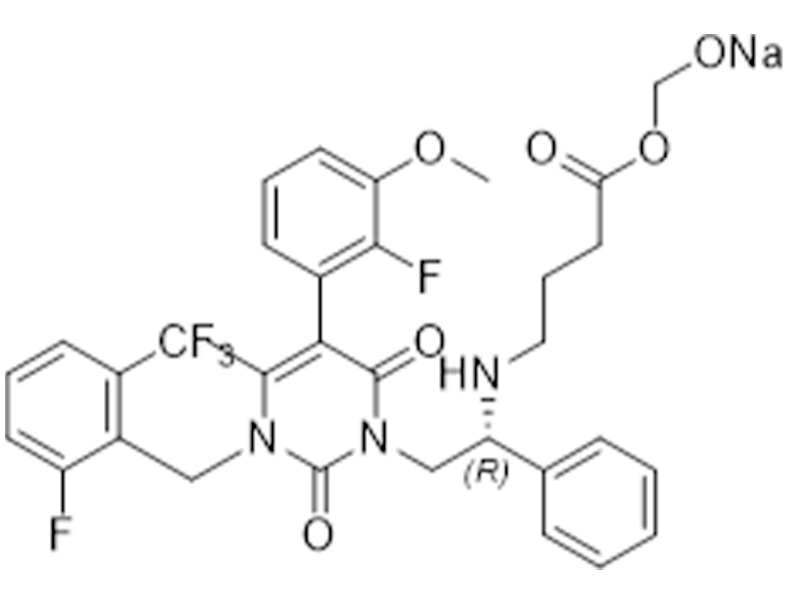

832720-36-2
企标
子宫内膜异位症
商业化/验证完成
恩格列净
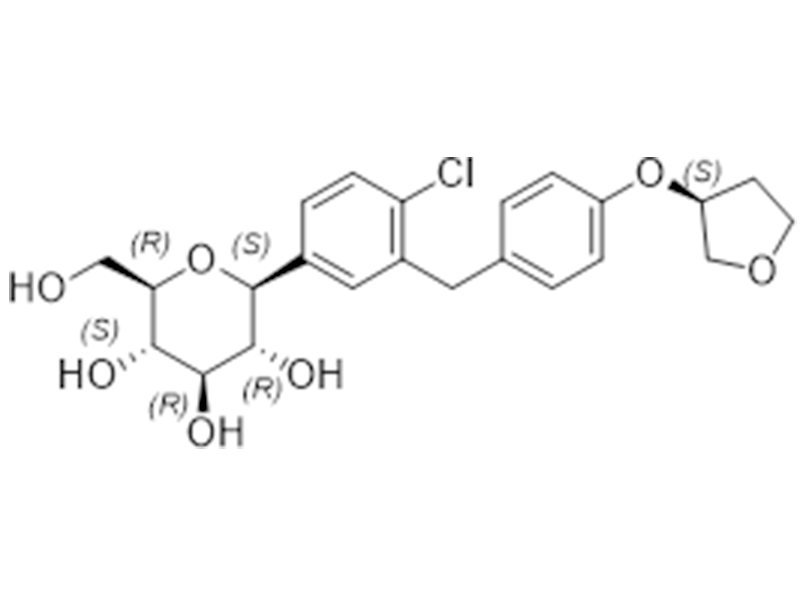

864070-44-0
企标
SGL2抑制剂/抗糖尿病
商业化/验证完成
恩扎卢胺
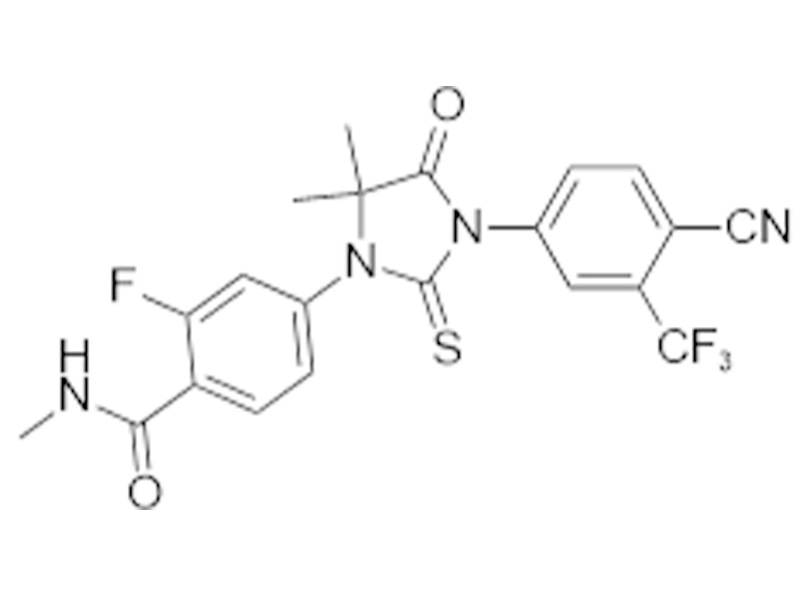

915087-33-1
企标
抗癌
商业化/验证完成
依折麦布
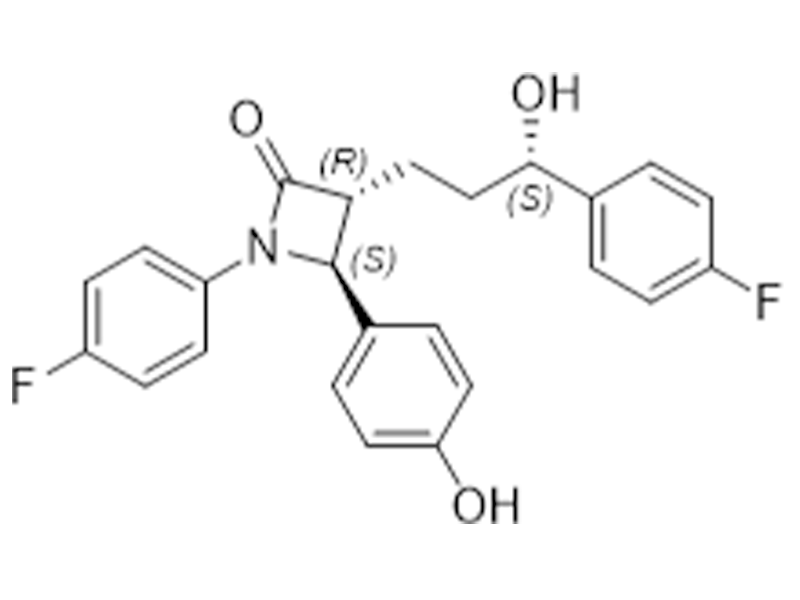

163222-33-1
USP
高胆固醇
商业化/验证完成
非布司他
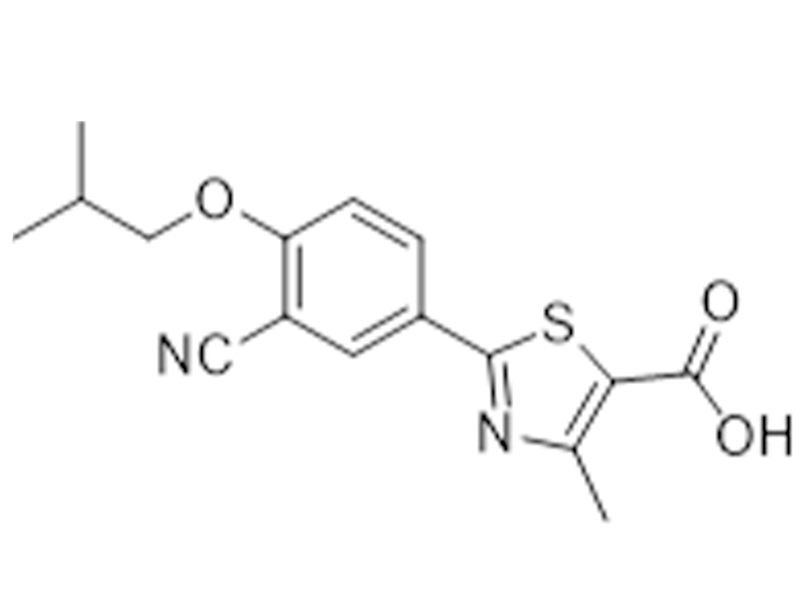

144060-53-7
企标
高胆固醇,痛风
商业化/验证完成
伊布替尼
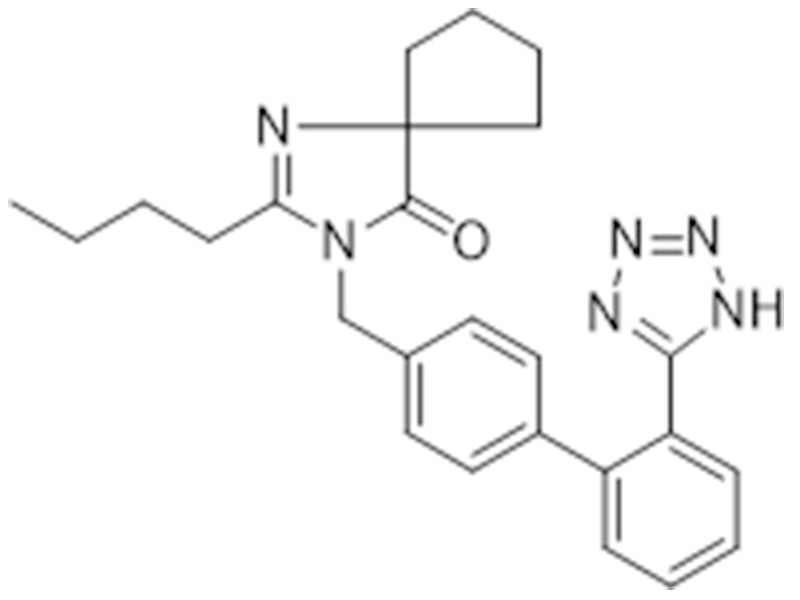

138402-11-6
企标
抗癌
商业化/验证完成
厄贝沙坦
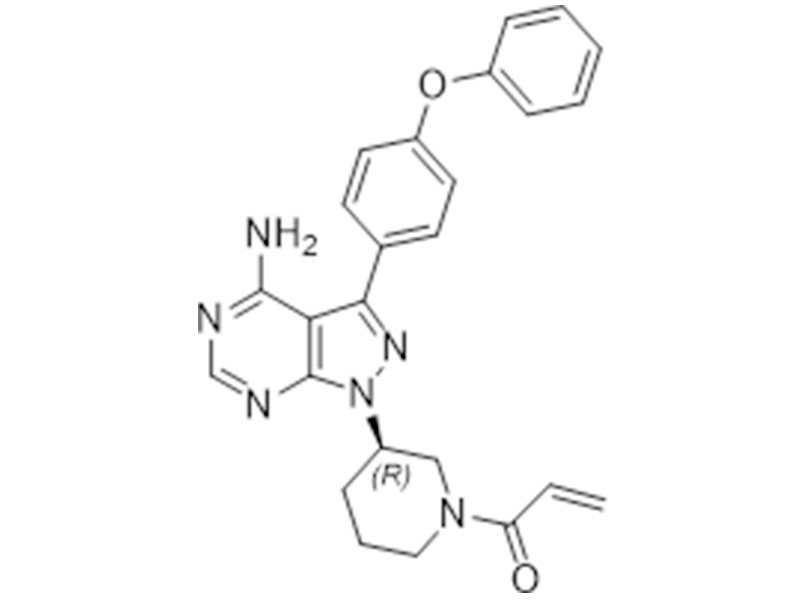

936563-96-1
USP/EP
抗高血压
商业化/验证完成
氯沙坦钾
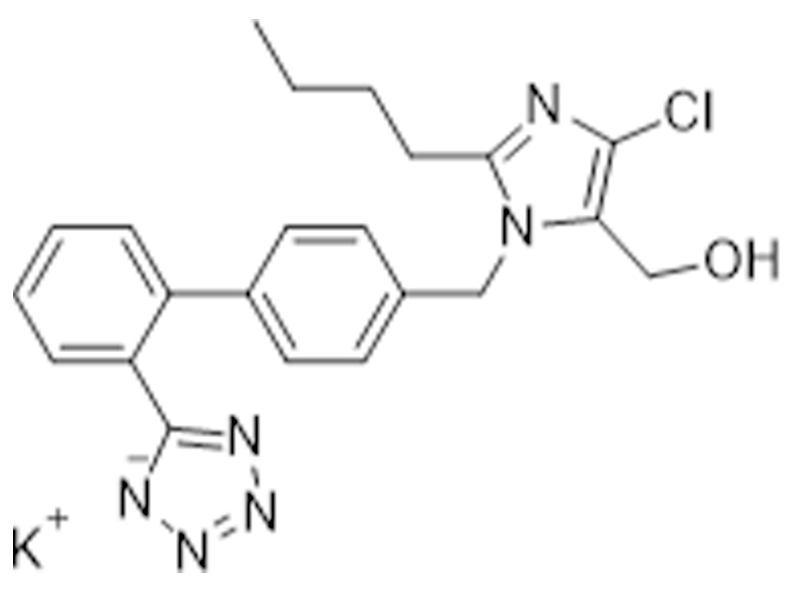

124750-99-8
USP/EP
抗高血压
商业化/验证完成
孟鲁司特钠
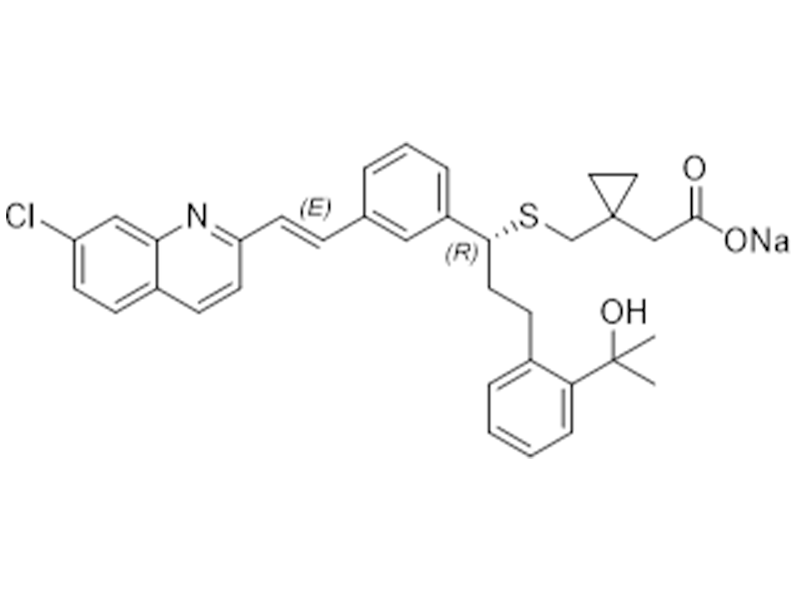

151767-02-1
USP/EP
抗哮喘
商业化/验证完成
尼可地尔
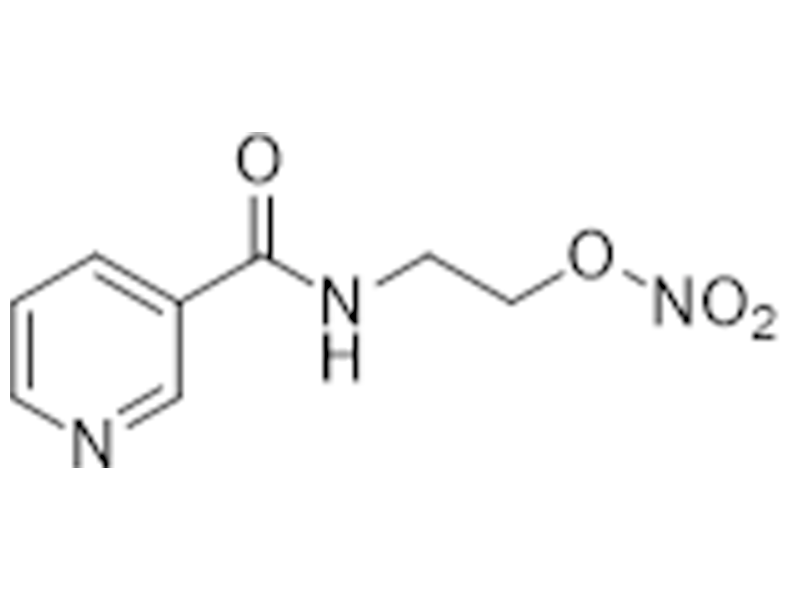

65141-46-0
JP/EP
抗心绞痛
商业化/验证完成
奥美沙坦酯
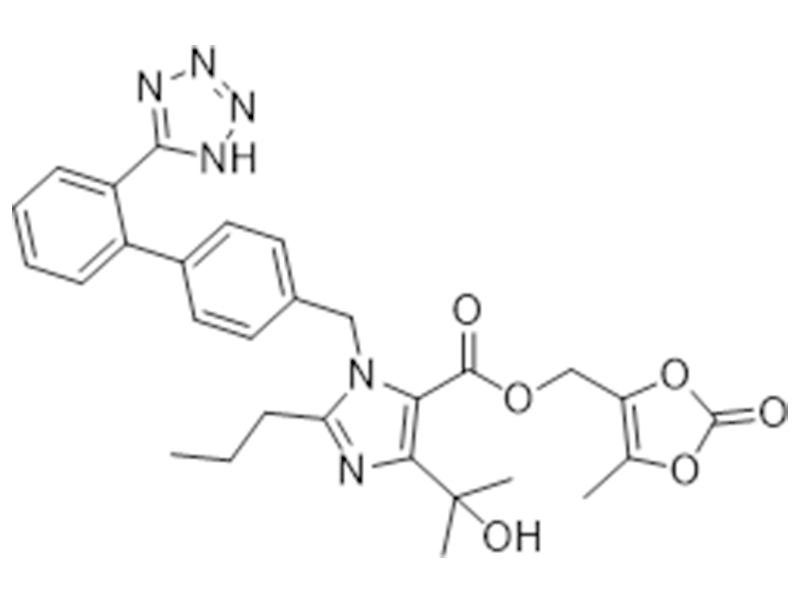

144689-63-4
USP/EP
抗高血压
商业化/验证完成
普瑞巴林
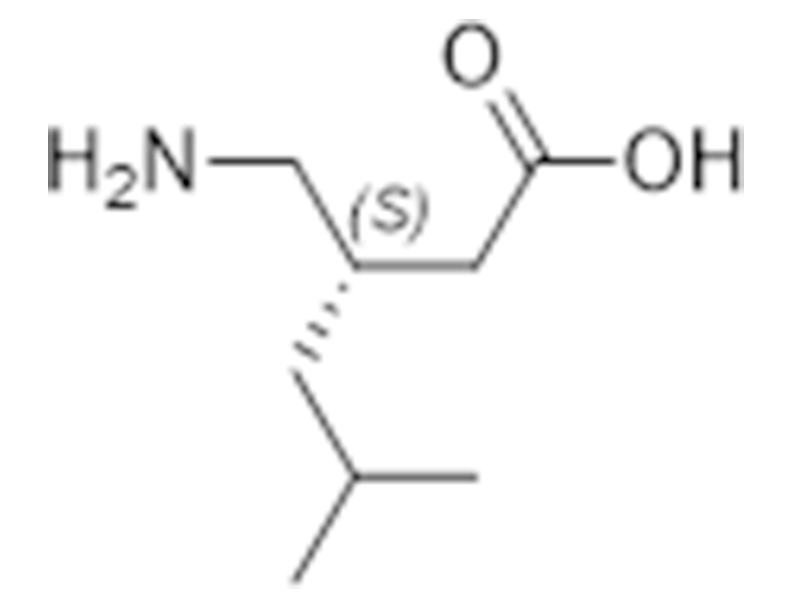

148553-50-8
企标
抗癫痫
商业化/验证完成
利伐沙班
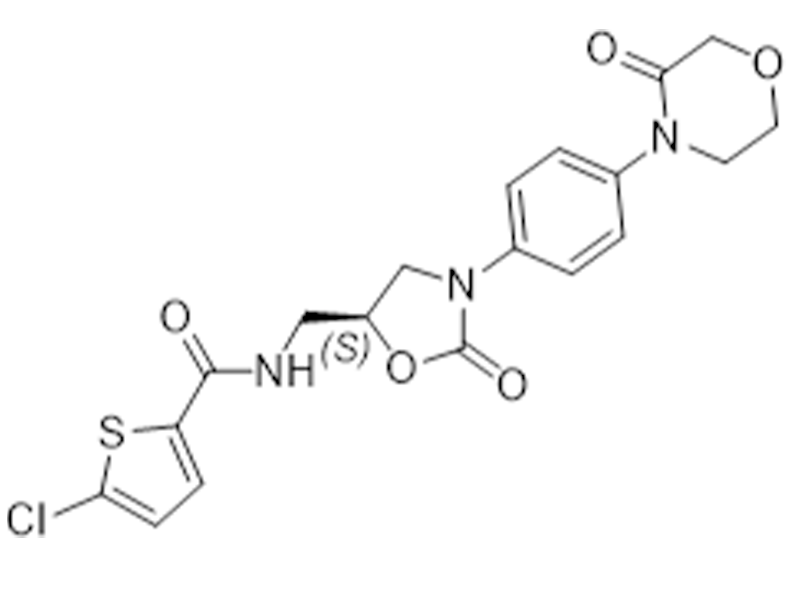

366789-02-8
EP
抗凝血
商业化/验证完成
沙库巴曲缬沙坦钠
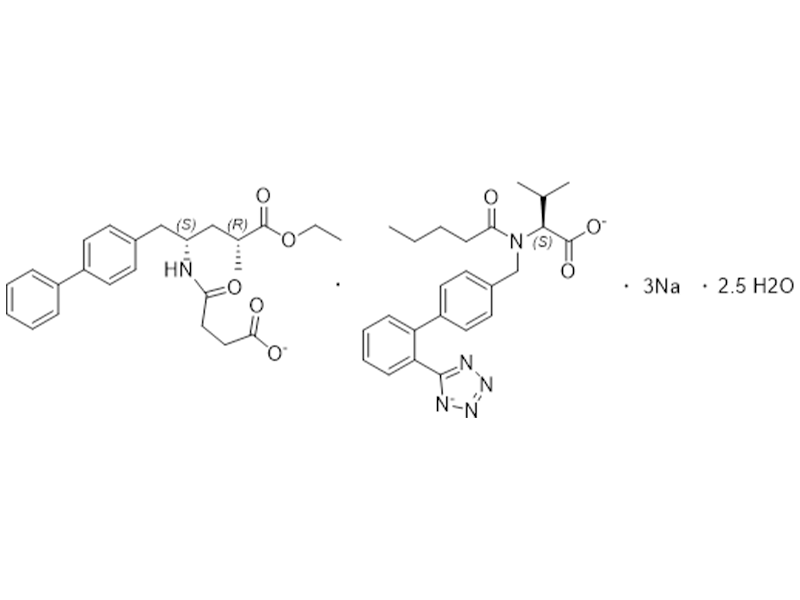

936623-90-4
企标
抗高血压
商业化/验证完成
赛洛多辛
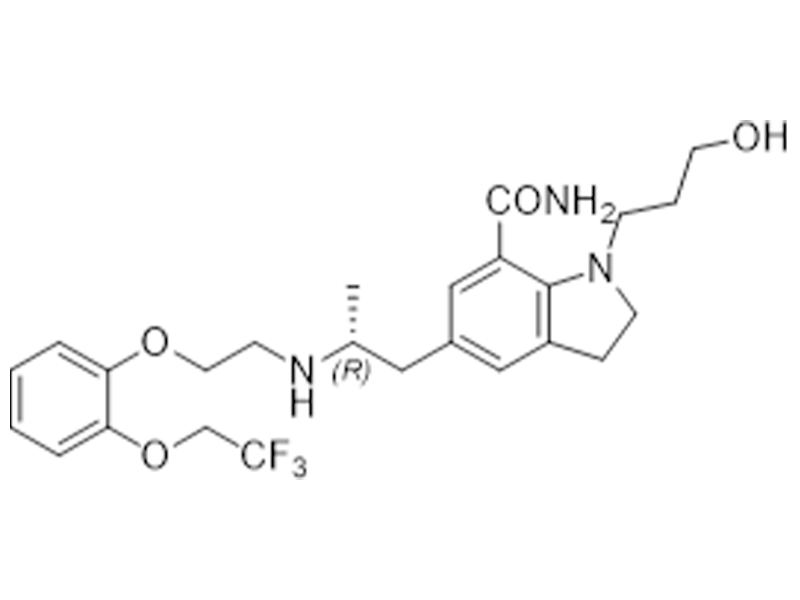

160970-54-7
JP/In house
良性前列腺增生
商业化/验证完成
盐酸西格列汀一水合物
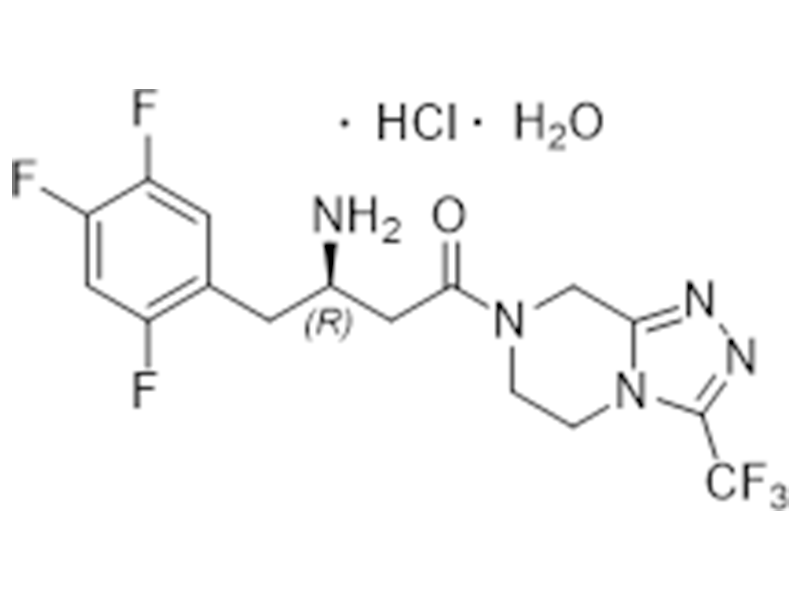

862156-92-1
企标
抗糖尿病
商业化/验证完成
磷酸西格列汀一水合物
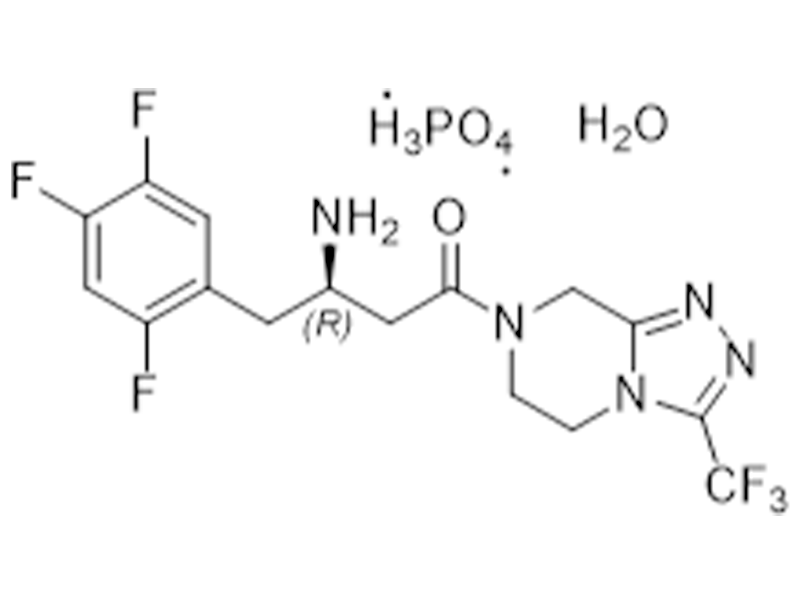

654671-77-9
USP/EP
抗糖尿病
商业化/验证完成
他氟前列素
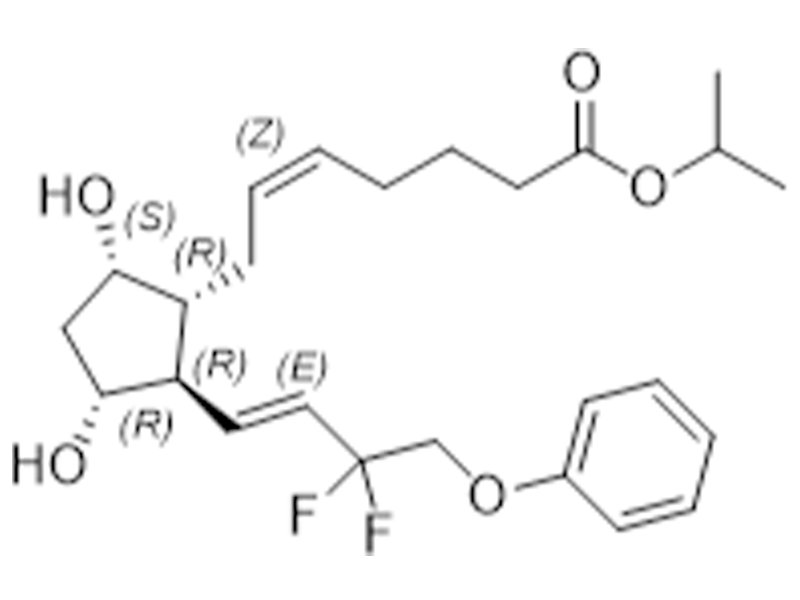

209860-87-7
企标
青光眼
商业化/验证完成
替米沙坦
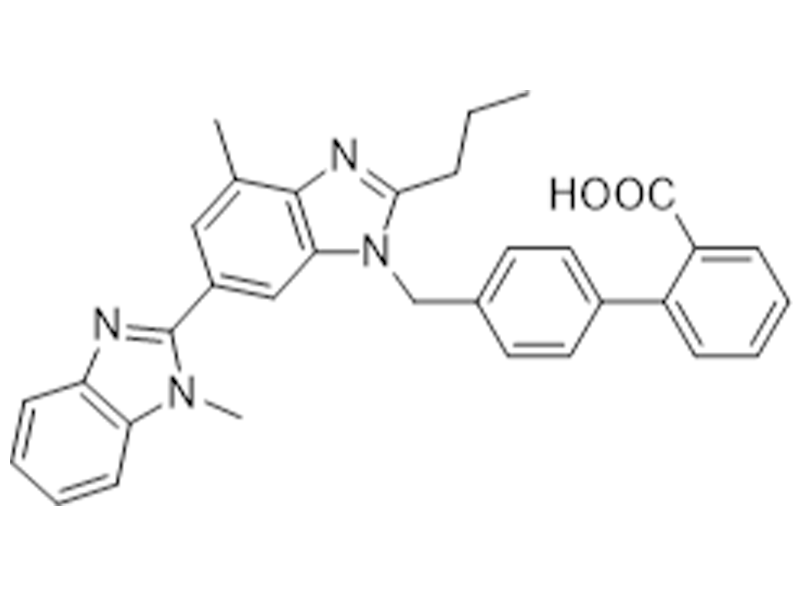

144701-48-4
USP/EP
抗高血压
商业化/验证完成
替格瑞洛
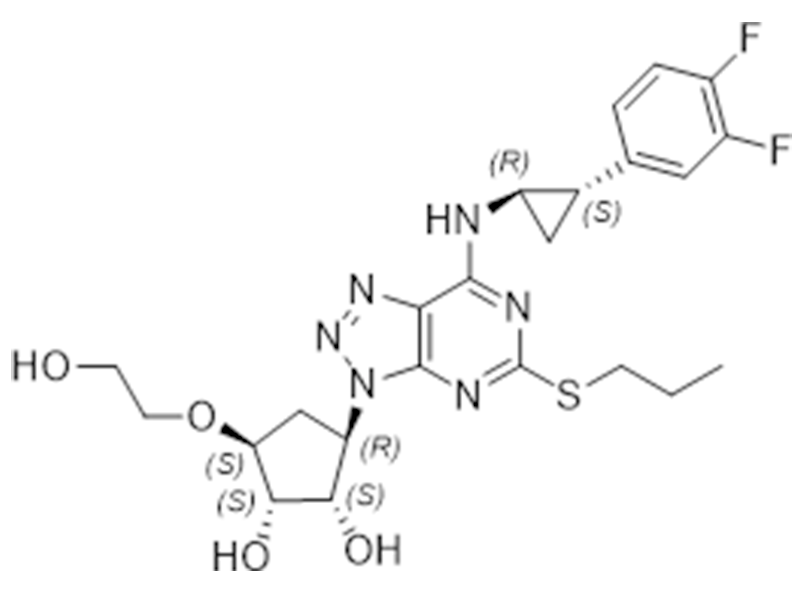

274693-27-5
EP
抗血小板
商业化/验证完成
缬沙坦
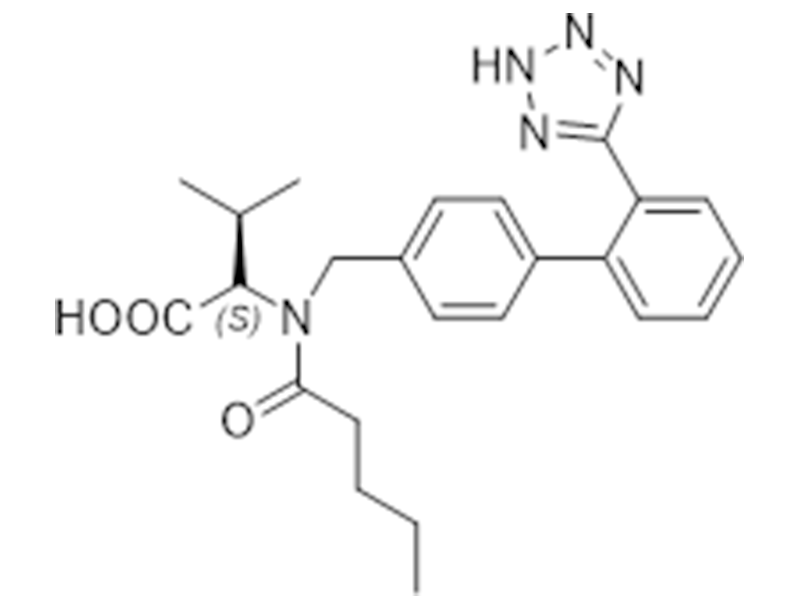

137862-53-4
USP/EP
抗高血压
商业化/验证完成
维格列汀
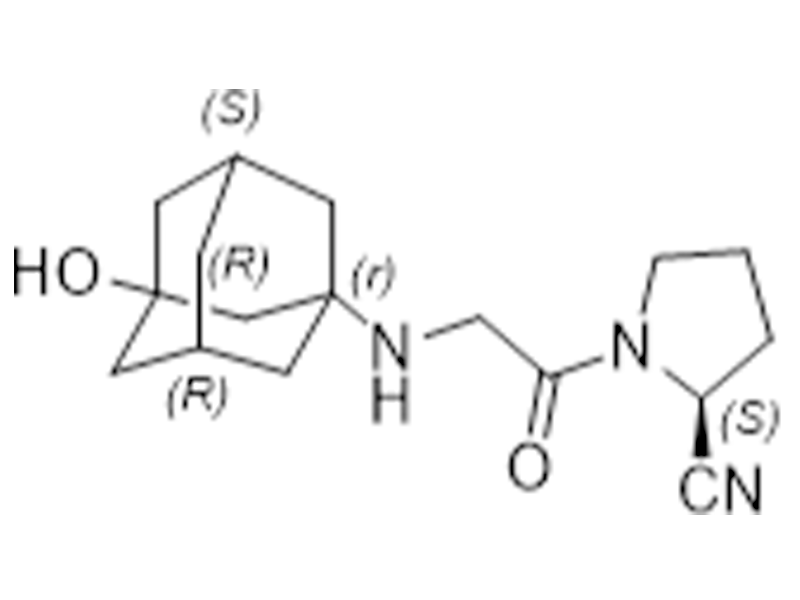

274901-16-5
企标
抗糖尿病
商业化/验证完成
富马酸伏诺拉生
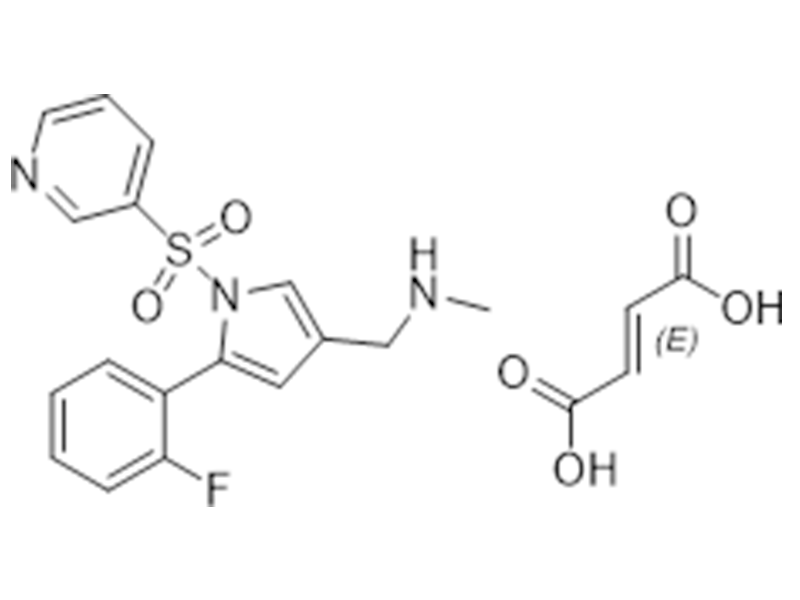

881681-01-2
企标
抗溃疡
商业化/验证完成
达格列净游离碱
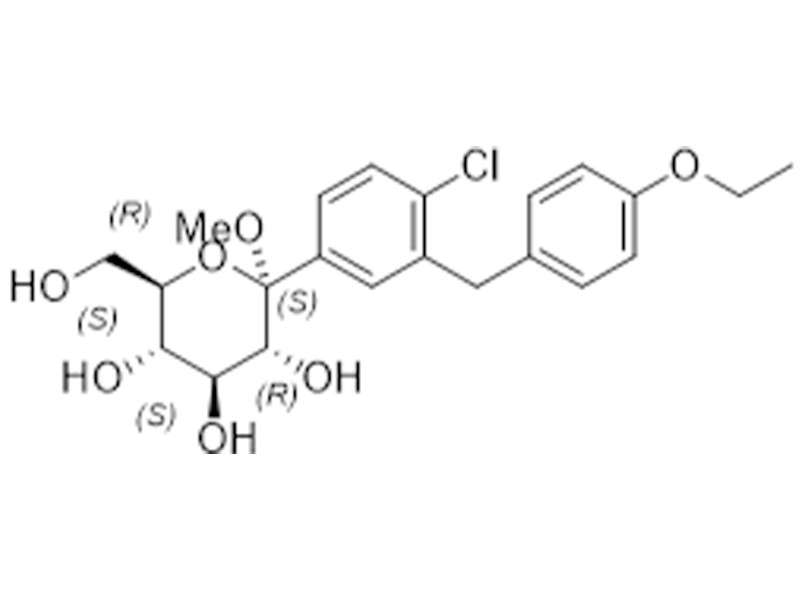

461432-26-8
企标
抗糖尿病
商业化/验证完成
瑞卢戈利
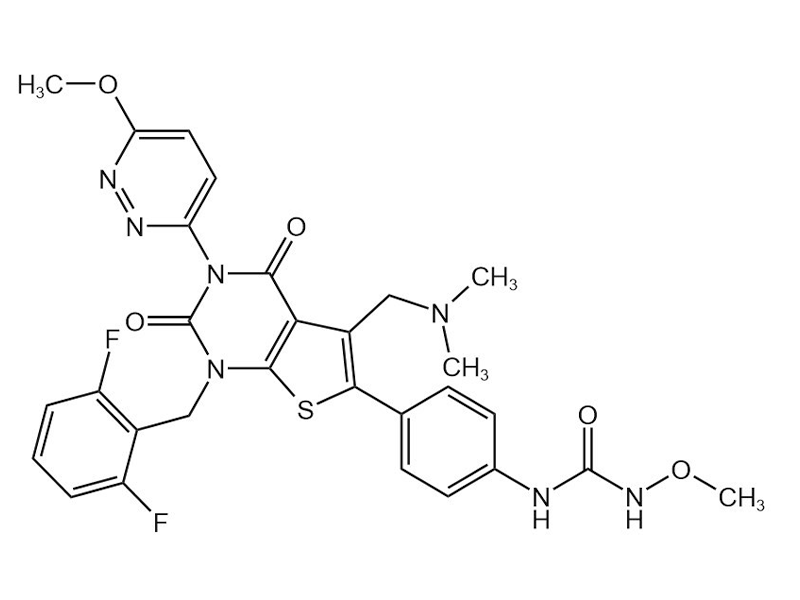

737789-87-6
企标
子宫内膜异位症
商业化/验证完成
比拉斯汀
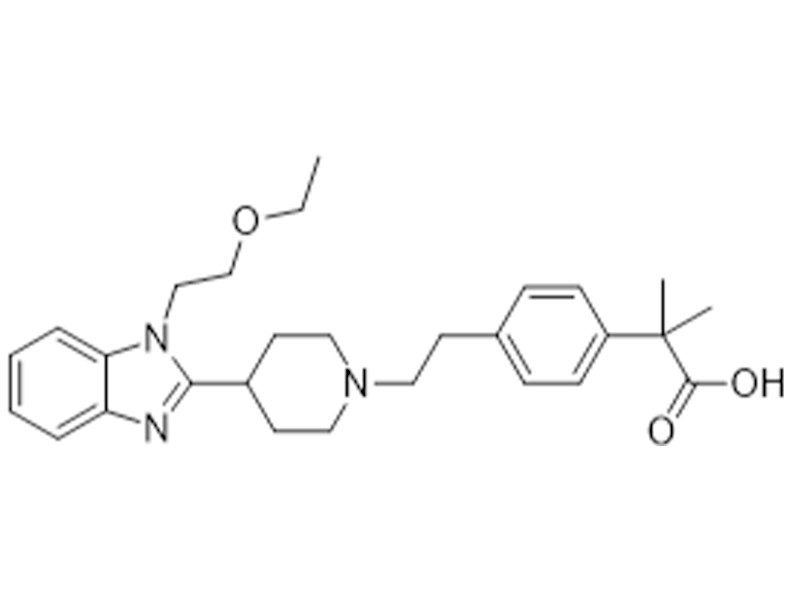

202189-78-4
企标
抗组胺药
商业化/验证完成
盐酸奈必洛尔
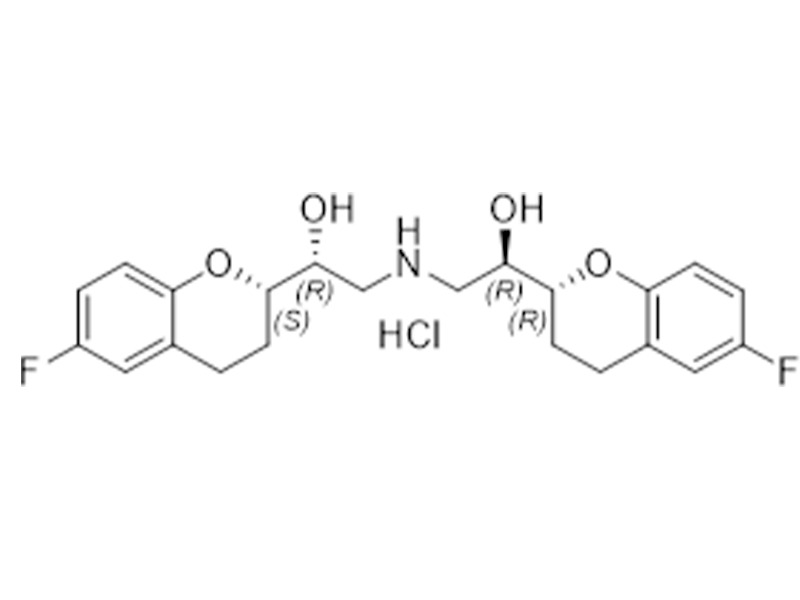

152520-56-4
CEP
抗高血压
商业化/验证完成
维贝格隆
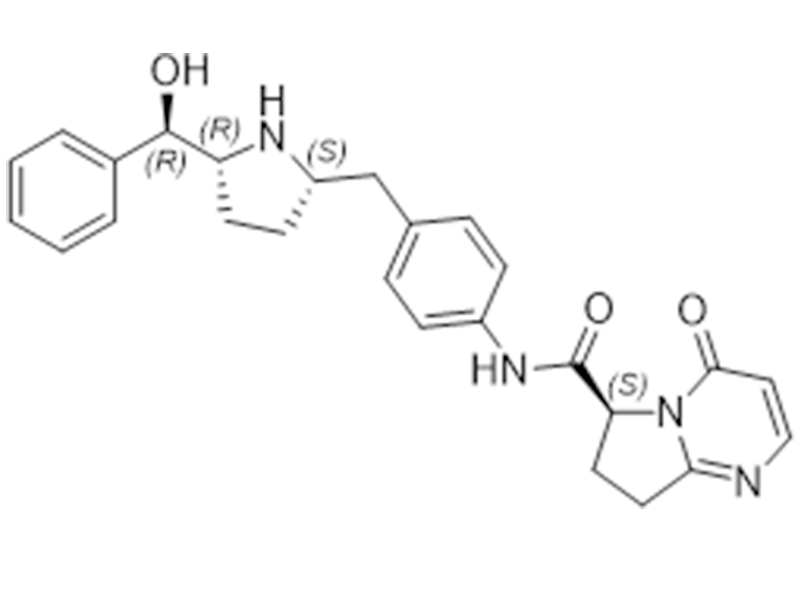

1190389-15-1
企标
膀胱过度活动
商业化/验证完成
阿普昔腾坦
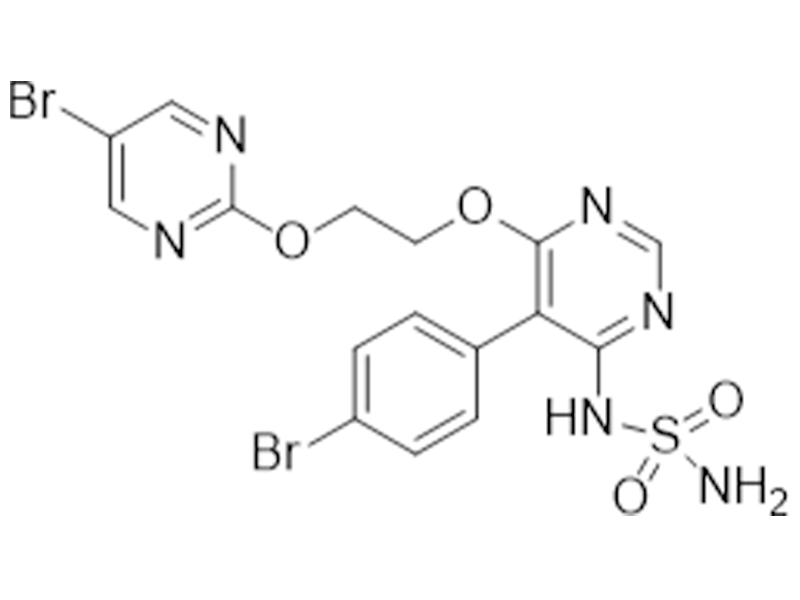

1103522-45-7
企标
抗高血压
商业化/验证完成
贝派度酸
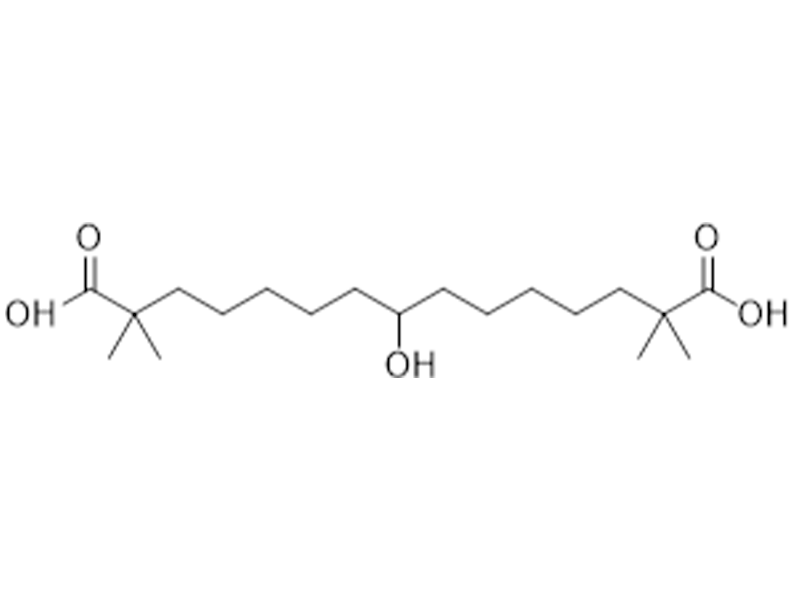

738606-46-7
企标
降血脂药
商业化/验证完成
非奈利酮
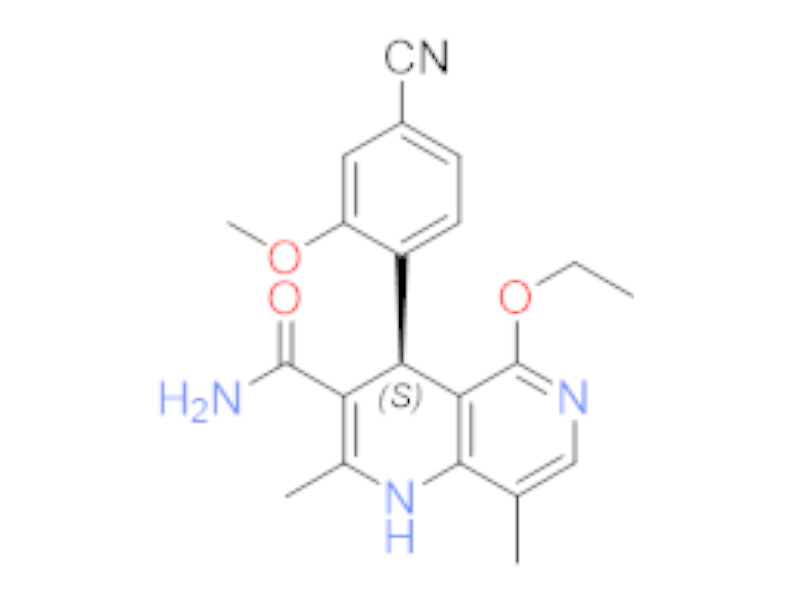

1050477-31-0
企标
糖尿病肾病
商业化/验证完成
联系我们





